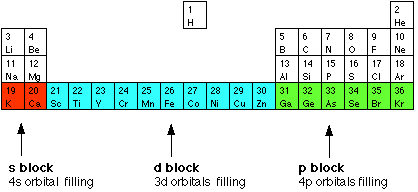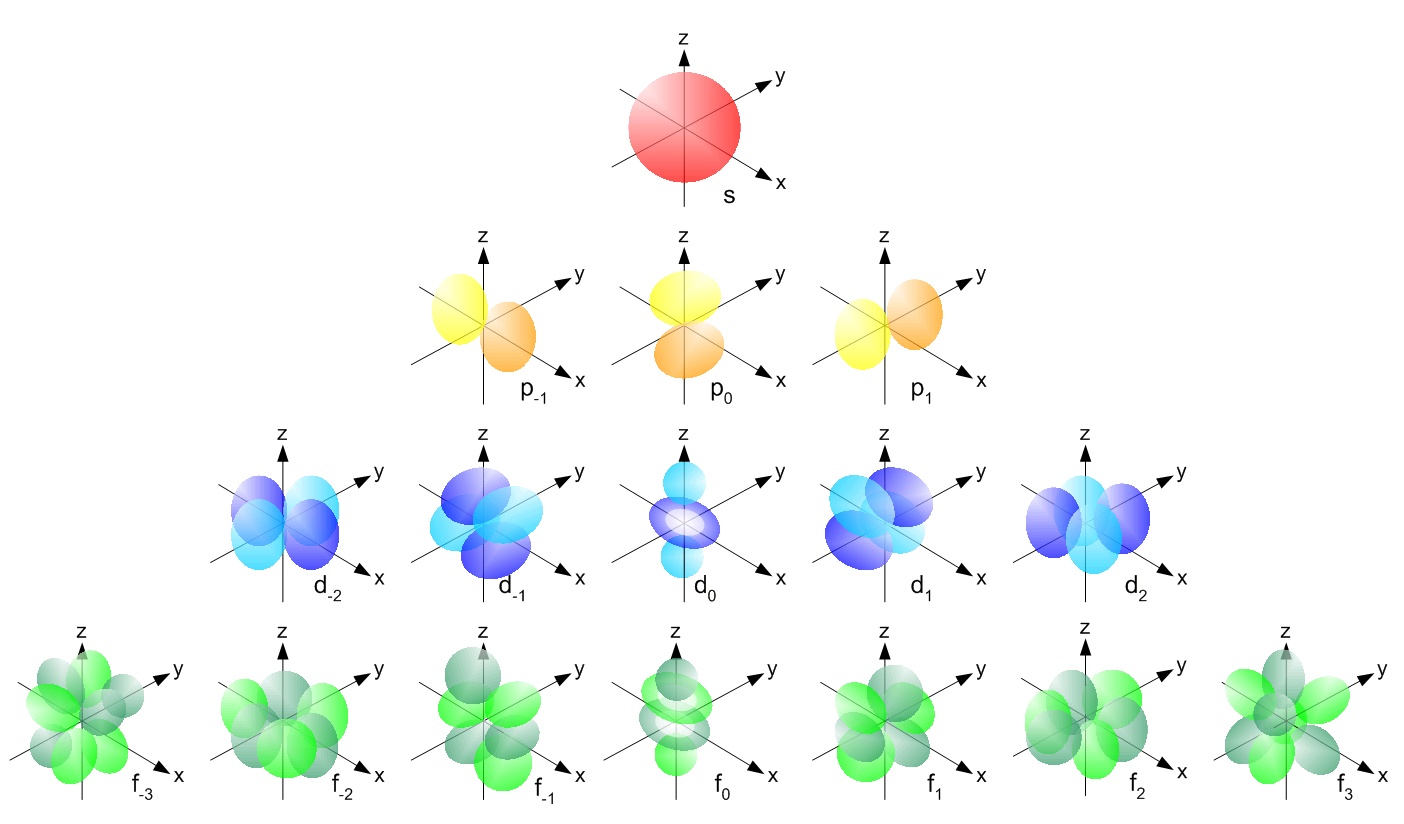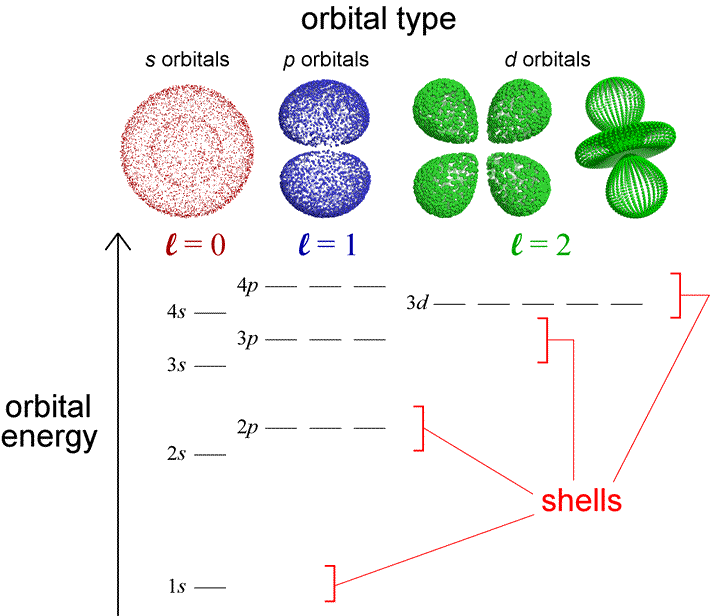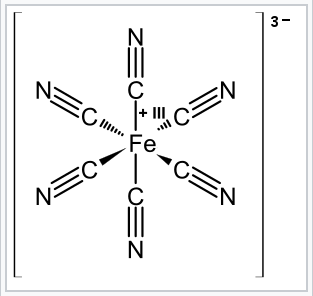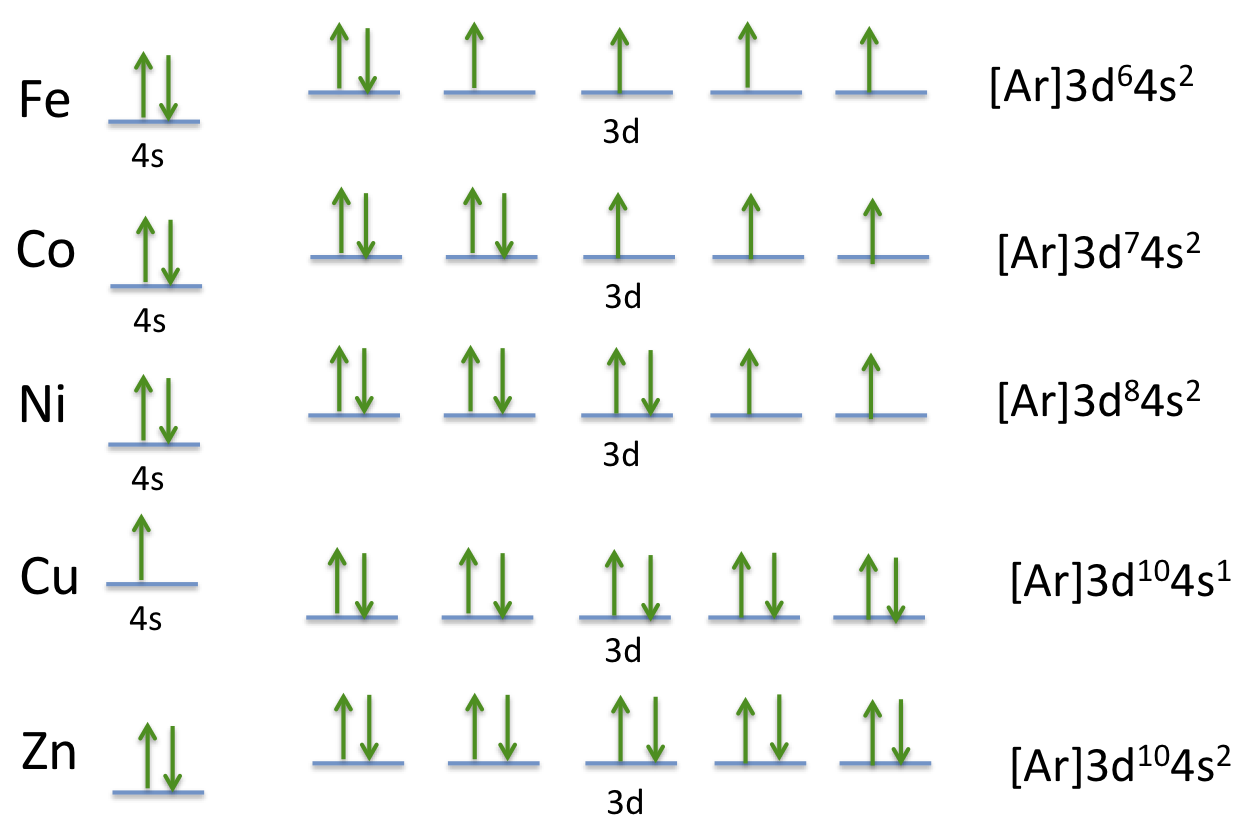
How many unpaired electrons are in an atom of Co in its ground state ? A) 7 B) 1 C) 3 D) 2 | Socratic

The d-electron configurations of `Cr^(2+), Mn^(2+), Fe^(2+)` and `Co^(2+)` are `d^4, d^5, d^6` and `d^7` respectively. Which one of the following will - Sarthaks eConnect | Largest Online Education Community