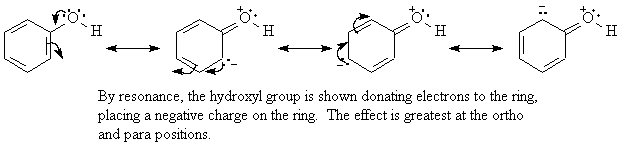Substrate scope. Electron-withdrawing and electron-donating as well as... | Download Scientific Diagram

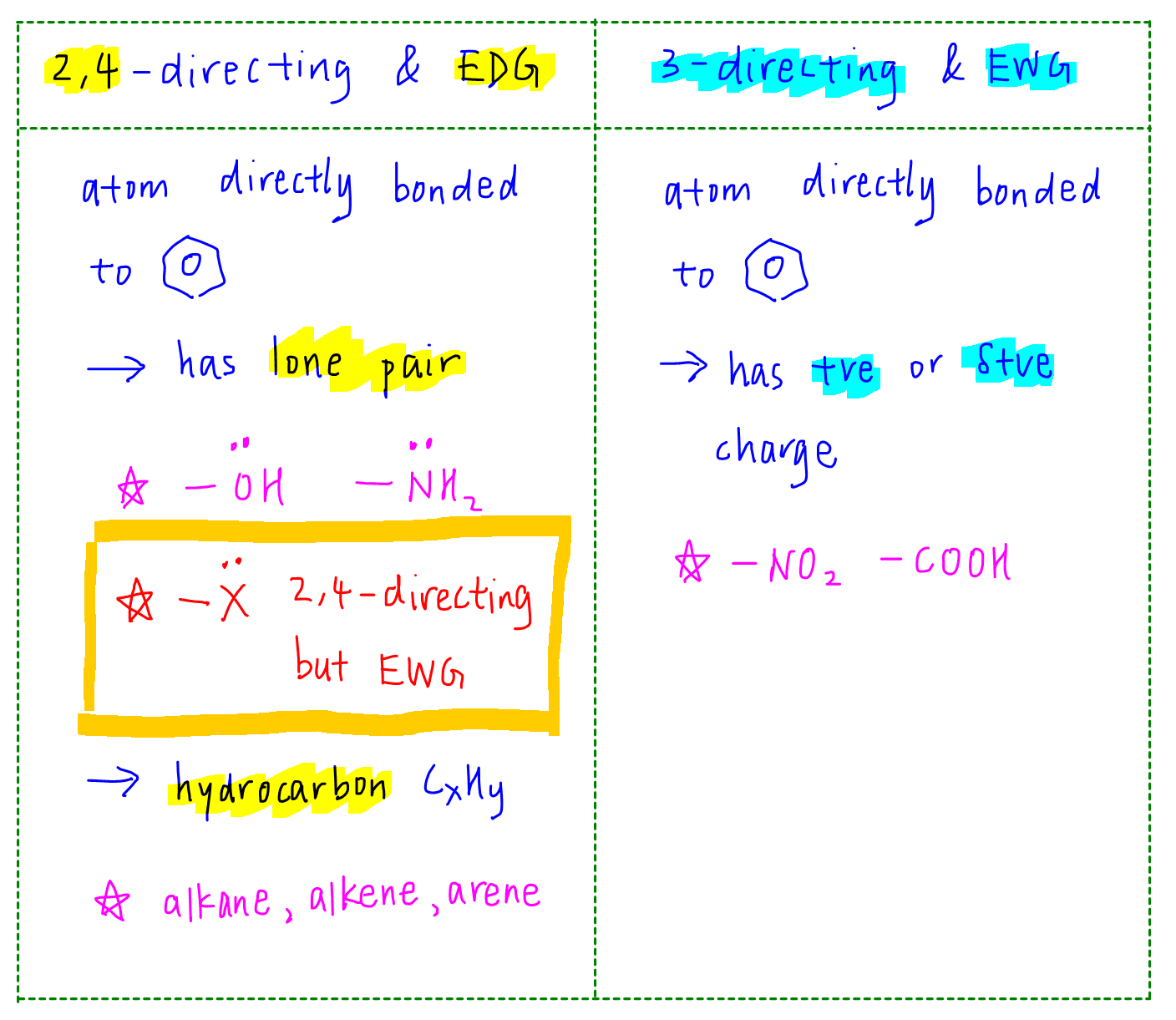
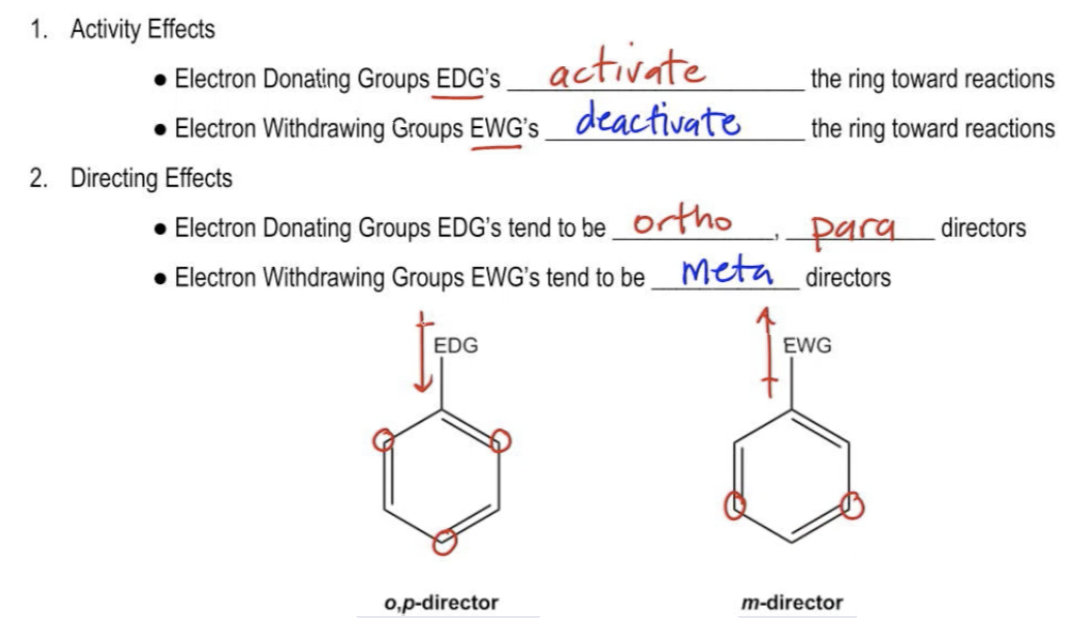
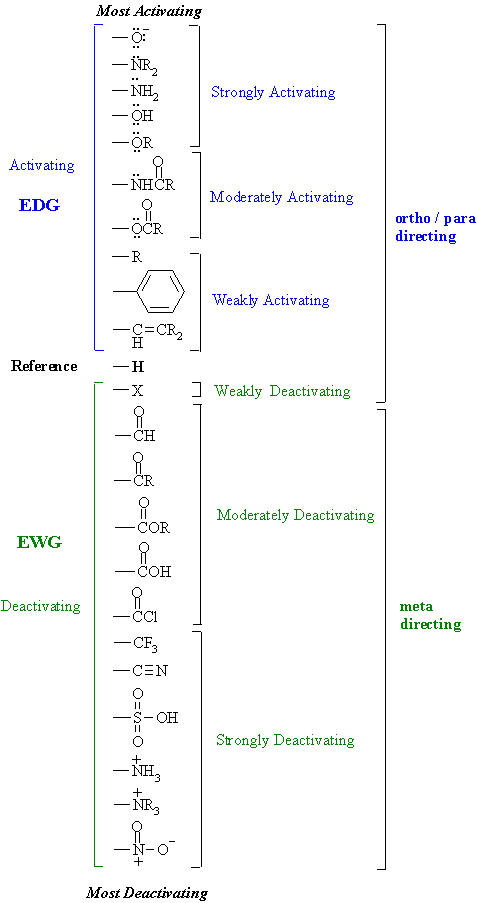
Why are ortho and para directors in aromatic rings electron donating whereas meta directors are electron withdrawing? - Quora

Substituent, structural and positional isomerisation alter anti-oxidant activity of organochalcogen compounds in rats' brain preparations - ScienceDirect
![Study the following sequence of reactions and identify the product (Y) . CH3CHO + HCHO [heat]dil.NaOH X [H3O^ + ]HCN Y Study the following sequence of reactions and identify the product (Y) . CH3CHO + HCHO [heat]dil.NaOH X [H3O^ + ]HCN Y](https://dwes9vv9u0550.cloudfront.net/images/2140650/bcc67e75-4fb0-4420-aaef-47cd4d3dbd3e.jpg)
Study the following sequence of reactions and identify the product (Y) . CH3CHO + HCHO [heat]dil.NaOH X [H3O^ + ]HCN Y

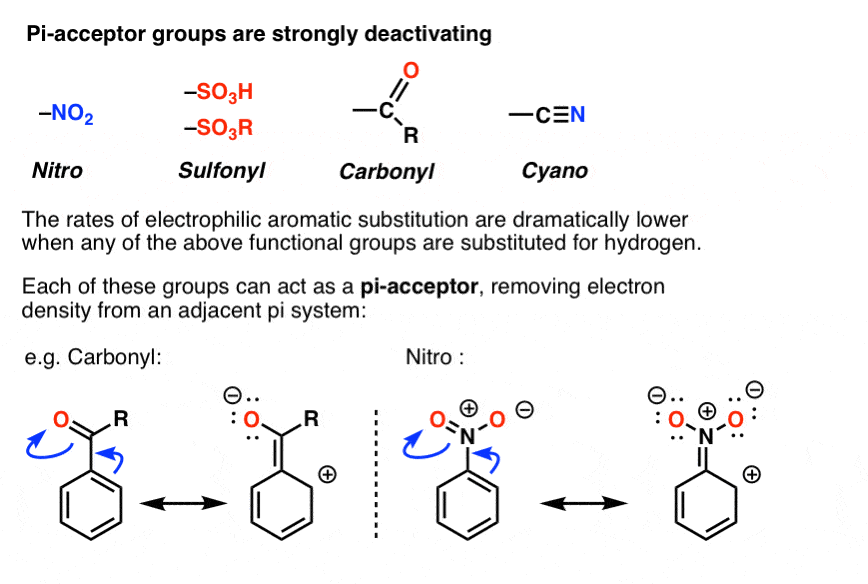
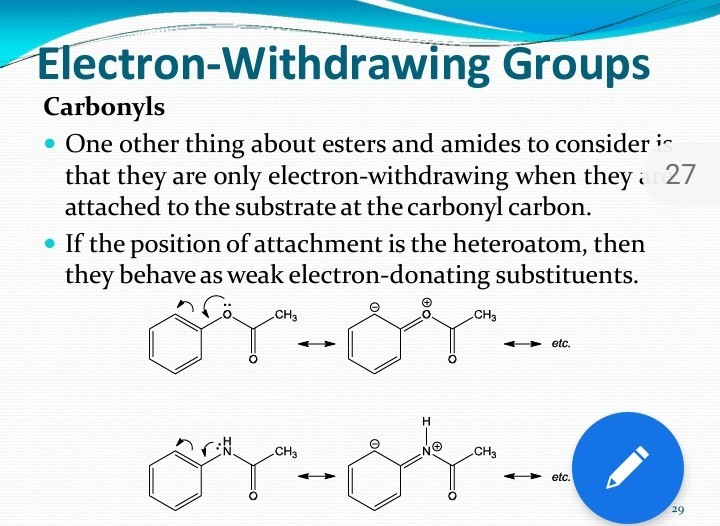
SOLVED: Electron-Withdrawing Groups Carbonyls When one of the atoms directly connected to the carbonyl carbon isa heteroatomas in esters and amides lone Pairh from the heteroatom can bepushed into the carbonyl group

Influence of the electron donor groups on the optical and electrochemical properties of borondifluoride complexes of curcuminoid derivatives: a joint ... - RSC Advances (RSC Publishing) DOI:10.1039/C6RA25436E

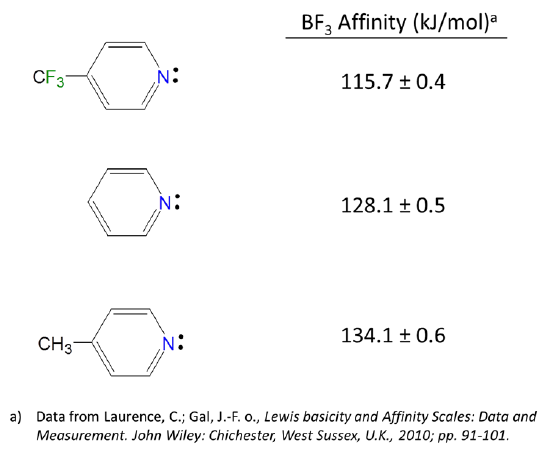
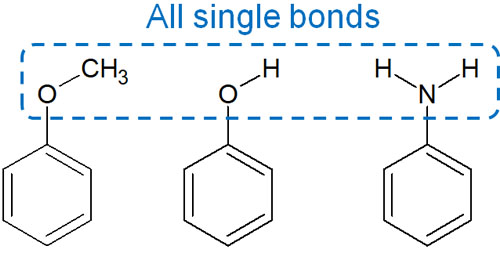
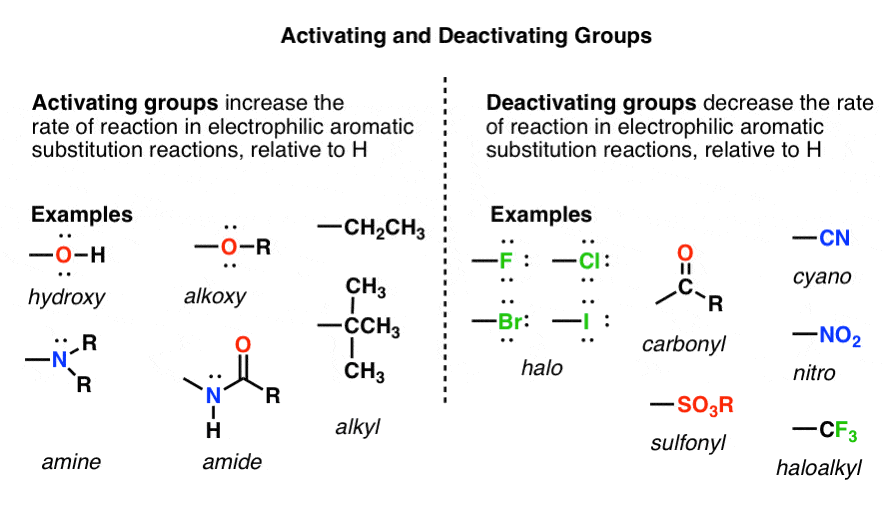
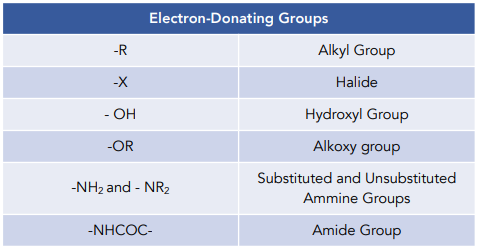
organic chemistry - Amine group - electron donating or withdrawing group? - Chemistry Stack Exchange