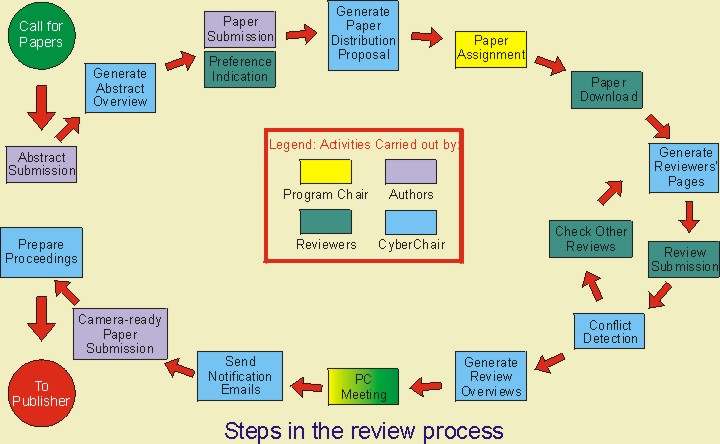
On 07.05.2019, the CMDE (Center for Medical Devic Evaluation), announced the introduction of NMPA's new eRPS System (Electronic Regulated Product

FDA Draft Guidance on Electronic Submission Template for Medical Device 510(k) Submissions | RegDesk

Holger Grünewald (Jülich, Germany) - Electronic Submission System and online Partner Search Tool - YouTube