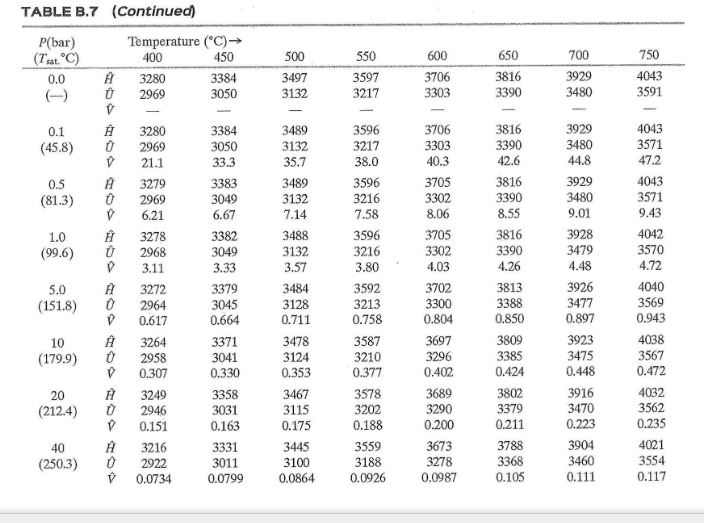
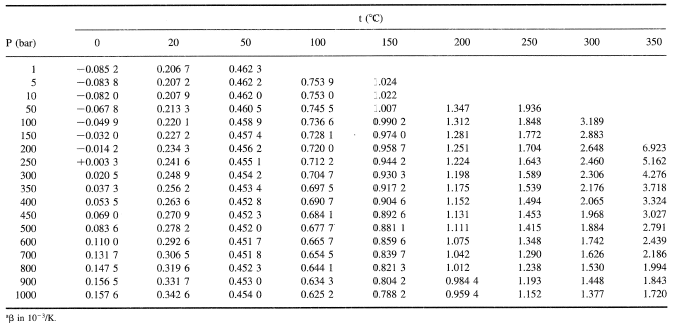
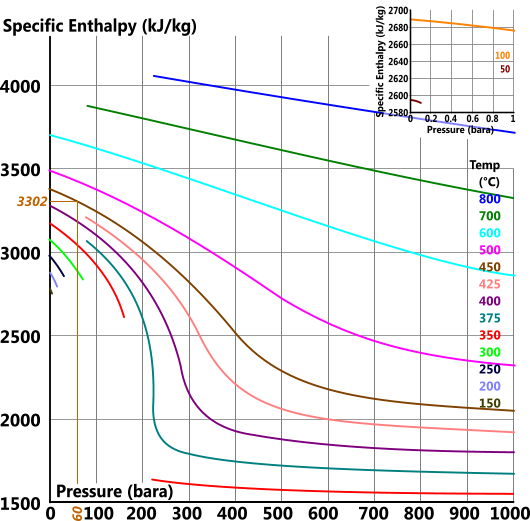
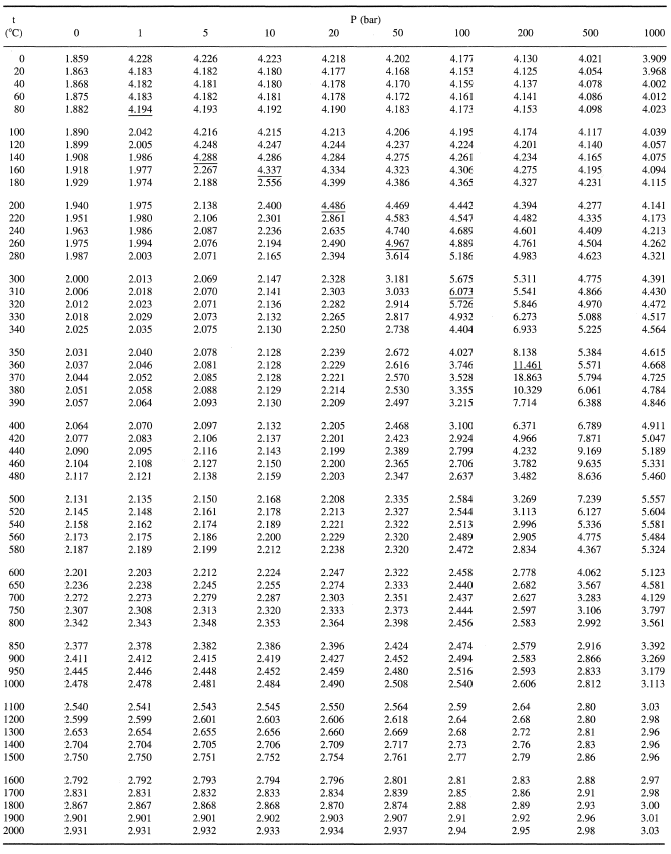
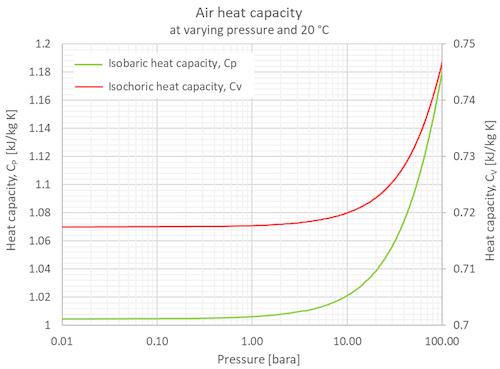
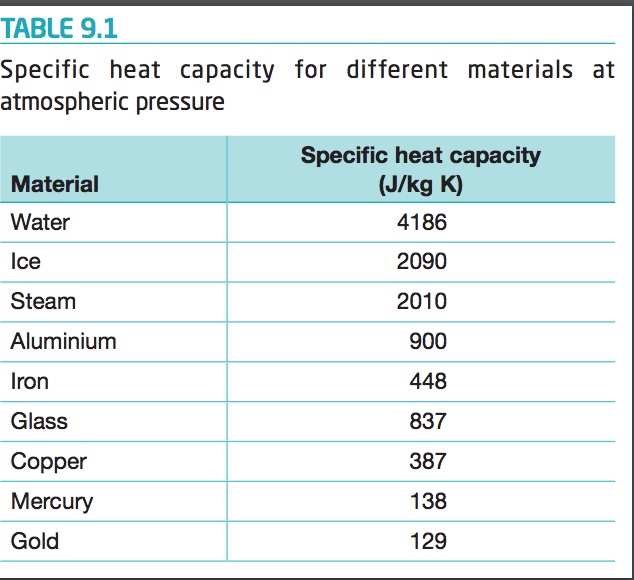
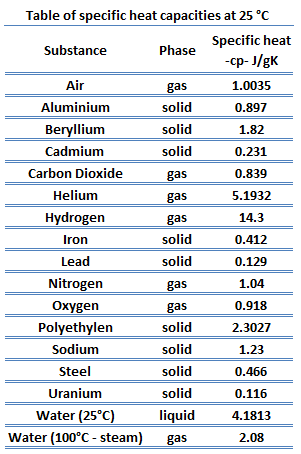
How many J of energy are needed to heat 45.0g of steam from 130oC to 245oC? Why don't you use 4.18 J/goC in this calculation? - Quora

SOLVED: Question 5 of 12 Submit What amount of heat, in kJ, is required to convert 1.20 g of water at 67.0 *C to 1.20 g of steam at 100.0 *C? (specific

The coefficients of the polynomial of the specific heat for water vapor. | Download Scientific Diagram
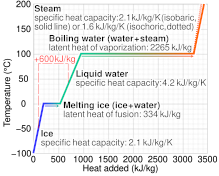
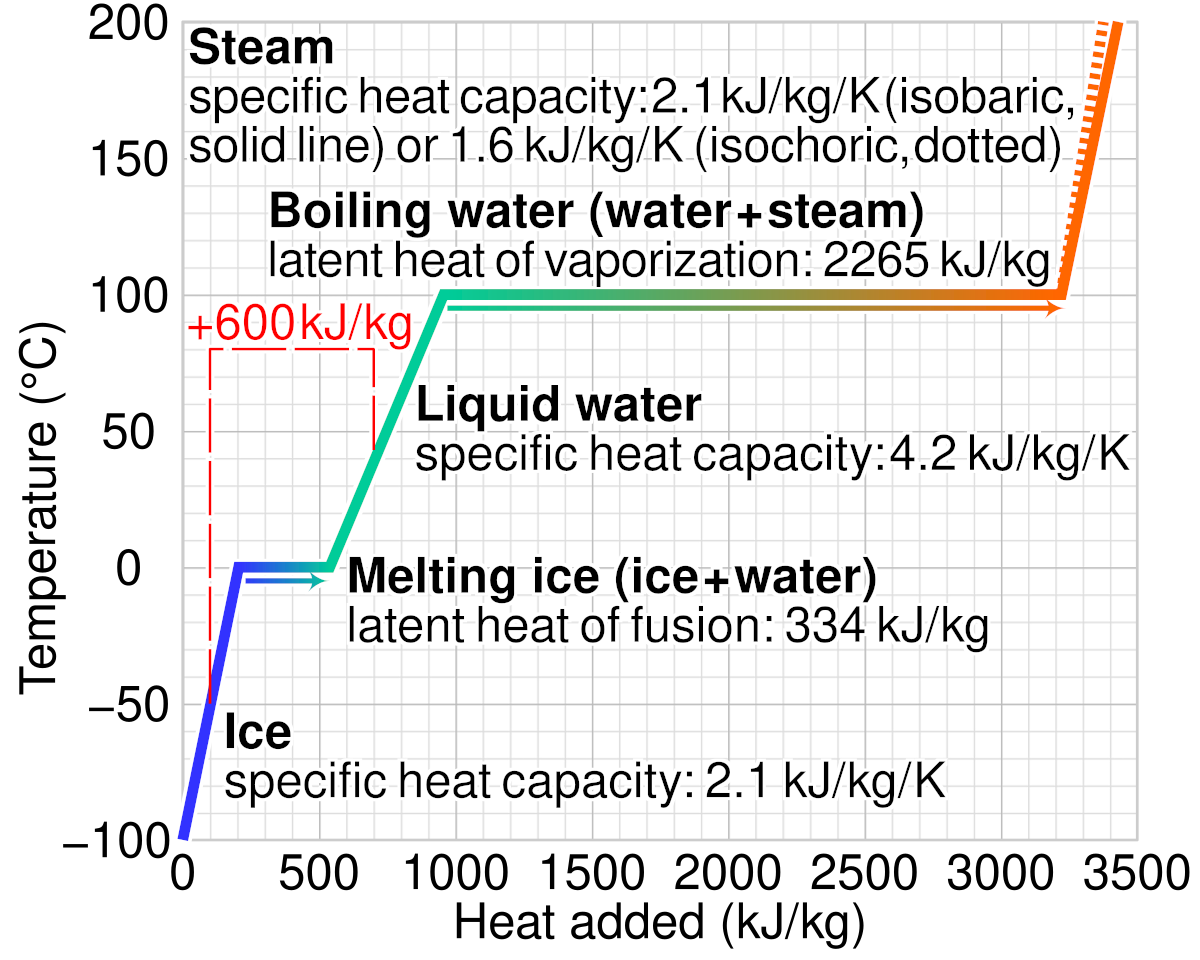
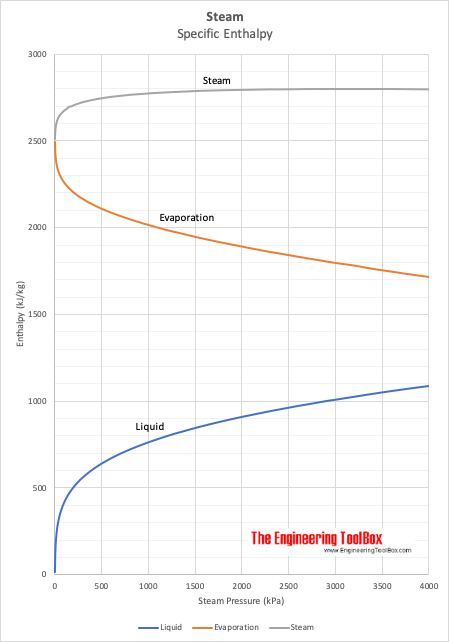
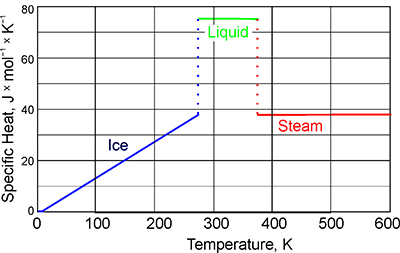
What are the values of the specific heat capacity of water, steam, and ice (in both joule and cal.) and the values of latent heat of fusion and vaporisation for water? -

Calculate the heat required to convert 3 kg of ice at `-12^()C` kept in a calorimeter to steam a... - YouTube
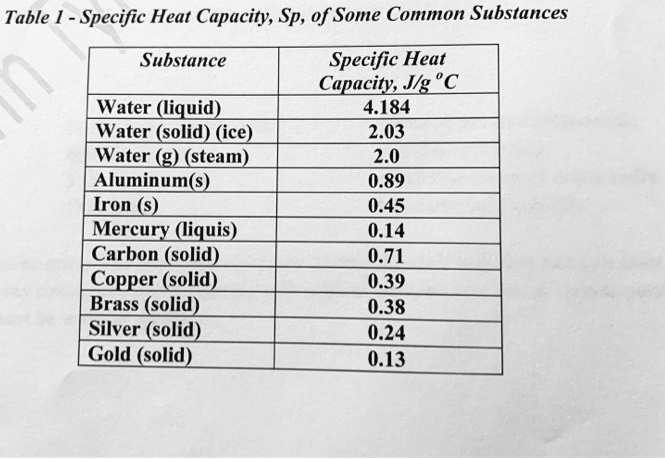
SOLVED: Table 1 Specific Heat Capacity, Sp, of Some Common Substances Substance Specific Heat Capacity, Jg 4.184 2.03 2.0 0.89 0.45 0.14 0.71 0.39 0.38 0.24 0.13 Water (liquid) Water (solid) (ice)

Specific heat capacity of superheated steam at 100 and 80 bar. Fig. 4.... | Download Scientific Diagram